Question 2a: Answer |
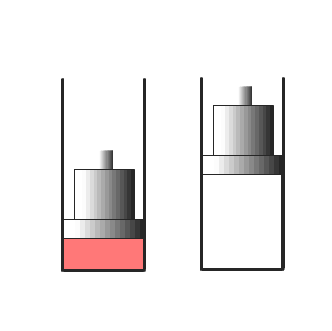 |
| The gas on the left is compressed adiabatically - the internal energy is raised by the work done on the piston. Slowly the heat escapes from the compressed gas. The gas on the right is compressed slowly allowing heat to escape to the surroundings. The temperature remains close to room temperature. |