The density of a 50% water/ethanol solution
Research question
What is the density of a 50 % water/ethanol solution?
Hypothesis
The density of water at room temperature is given as 1000 kg m-3 and the density of ethanol is given as 800 kg m-3 (MAOL's tables). It is expected that the density of a 50% water/alcohol solution will be 900 kg m-3, half way between the densities of water and alcohol.
Apparatus and procedure
Density r is given by Mass/Volume.
Five clean 100 ml graduated measuring cylinders, were placed,
one at a time, on an electronic balance, accurate to within ±.0.001 g, and filled to the 50 ml level.
The mass before and after filling was noted. Room temperature
was measured to within ±1°C with a mercury in glass
thermometer. The solution and cylinders were left in the room
(out of direct sunlight) for 12 hours to ensure uniform temperature.
Data
Table 1 lists the measured mass of 50 ml of solution (mass full
- mass empty) in each cylinder.
Table 1
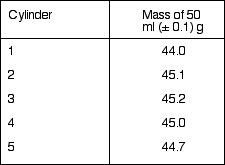
The average mass is 44.8.±.0.6 g ... where the error is half the range. Note: the balance was accurate to ±.0.001 g but the volume added to the cylinders was measured to within ±.0.5 ml. The temperature of the solution and the air temperature in the room were 21 °C, with an uncertainty of .1°C.
Analysis
The density of the 50% solution is given by ...
Discussion
The density of the solution is 900 g/cc as expected, within ±1%.
Evaluation
The method was not as accurate as had been expected, because
it was not possible to pour exactly 50 ml of solution into the
cylinders. Future work would be more accurate if a nominal 50
ml was poured into a clean dry cylinder, and the volume read
on the scale, to the bottom of the meniscus, to within ±0.1
ml. A single cylinder could be calibrated for volume by repeatedly
finding the known density of water at a measured temperature.
This would reduce any systematic errors due to an inaccurate
volume scale.
|
Editor's comments The title is now improved. The student seems to have used a template supplied by the teacher. The numbers for sections add nothing and have been deleted. The serious flaws in the original work are now painfully apparent. Attempting to pour 50 ml of solution into a cylinder is silly. The cylinder is graduated for a reason. The volume should be read, to the bottom of the meniscus, to the nearest tenth of a ml each time. Using five different cylinders may replace a systematic error with an additional random error. One cylinder could have been calibrated carefully using the known (published) density of distilled water at room temperature (21°C). That one cylinder could then have been used too measure the volume of a nominal 50 ml of solution five (or more) times. Washing the cylinders with acetone to remove any residual water was a waste of time. Just dry them. The errors in volume are a one or two orders magnitude larger than any other errors. Using what the author calls the "maximum residual" as the error is unsound. The value appears to be an outlier. The average deviation (or the standard deviation) are preferred. To keep it simple, half the range would do, or, if the student is determined to use residuals, the average deviation is acceptable. Calculations in the original appear to be wrong - and the detail in the error calculation is silly. Just do a double calculation and write down the error in density from the uncertainty in the volume measurement.
I doubt that the student actually made any measurements. I suspect they knew that the teacher wanted a density that was different from the 900 g/cc quoted, and they tried to make it up from there. Ed. |