Measurement Skills
Science is a quantitative subject. This means taking measurements and dealing with numbers.
- All measurements consist of two parts: the magnitude of the measurement (the number) and the unit of the measurement.
- Two types of quantities can be measured: scalar quantities and vector quantities. Scalar quantities can be completely described by magnitude and unit. Scalar quantities include mass, time and temperature. Vector quantities additionally require direction to completely describe a measurement. Examples of vector quantities are force and velocity.
- Measurements are never exact. In other words, measurements always contain a degree of uncertainty.
Significant Figures (Digits)
- The precision of lab measurements is indicated by the variation in a number of many repeated measurements. The more variation, the lower the precision of the measurement (this tells us how consistent we are when making measurements).
- Precision is also defined by resolution of the measuring instrument. A more “precise” instrument will provide readings with a more digits after the decimal point.
- Significant figures in a measurement include all the digits that are known for “certain” plus one more “uncertain” digit. The uncertain digit is the estimated digit in a measurement
- More precise instruments mean your measurements will have more significant figures (and smaller uncertainties)
How many significant figures are in a measurement?
In any given measurement, all non-zero digits (1 to 9) are always counted as significant. There are special rules for zeros when counting significant figures.
Rule |
Examples |
Zeros between non-zero digits are significant. |
40.5 has 3 S.F. |
Zeros following a non-zero digit and after a decimal are significant. |
5.230 has 4 S.F. |
Zeros before the first non-zero digit are not significant. A single zero used as a place marker before the decimal is NOT significant. |
0.000450 has 3 S.F. |
Zeros at the end of a number that does not have a decimal are not significant. |
100 has 1 S.F. or 3 S.F. |
Here is Practice Worksheet 2.1 on Significant Figures.
Measurements and Uncertainty
How long is the pencil?
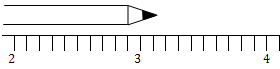
When making measurements, the last digit of the measurement is always an estimate. This digit is called the uncertain digit (because it is an estimate).
When taking a measurement:
- read the scale to the smallest graduation (the limit of measurement)
- estimate the next digit of the measurement
This produces a more precise measurement but with a degree of uncertainty. When deciding on a reasonable uncertainty in a measurement, one must consider the instrument itself AND how the measurement is made. A general starting point for an uncertainty determination is:
A rule of thumb: The degree of uncertainty is at a minimum equal to half of the smallest graduation on the measurement scale and is often greater.
Uncertainty and error are two very different concepts. An error in measurement is how far the measured quantity is from the actual value. Errors are not part of measurement uncertainty although discussion of errors is important in lab reporting.
The quantity of uncertainty reflects the precision of the measuring tool used (for example a 10 mL graduated cylinder is more precise than a 100 mL cylinder) AND any limitations in the measurement technique itself (for example, using a meter stick to measure the rebound height of a bouncing ball).
The National Institute of Standards and Technology (NIST) uses the symbol “u” to represent uncertainty. For a temperature measurement, the uncertainty would be represented as “uT”.
Example
|
 |
Key Concept: Notice that the uncertainty ± 0.5
- Has only one non-zero digit (one significant figure)
- The non-zero digit has the same place value as the estimated digit in the measured value.
This type of uncertainty is called an absolute uncertainty because it is measured in the same units as the measured quantity.
Now go back and write the length of the pencil and include it’s absolute uncertainty.
Relative uncertainty expresses the absolute uncertainty as a proportion (or percentage) of the measured quantity. For the previous measurement of T (temperature).
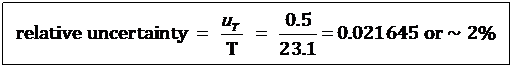
In ALL science labs in IB chemistry, you will never make a measurement without including an uncertainty. How you choose that uncertainty will depend on what the measurement tool was and how it was used. The uncertainty included will have to be reasonable. This will take practice and judgement.
Your estimates of uncertainty may be different than a classmate. It is important that your answer is reasonable and that the place value of the uncertainty is the same as that of the last digit of the measurement. A larger uncertainty reflects less confidence in the value of a measurement.
When using digital devices such as an electronic balance or a vernier temperature probe, the device may produce numbers that appear more accurate than the device is capable of measuring. For example, a Vernier temperature probe may detect a temperature that is recorded as 24.3235 °C in the data logger software. However if one looks in the manual, one finds that the uncertainty (sometimes referred to as the tolerance or readability) is 0.3 oC for temperatures from 5 to 90 °Celsius. So what should you do?
When an electronic device or data probe is used for a measurement, check the manual or check with the teacher what the uncertainty of the instrument is, then round values, appropriately, applying the uncertainty. In the example above we would round 23.3235 to 23.3 and write the final measurement as: 23.3 ± 0.3 °C.
Here is Practice Worksheet 2.2 on Measuring Uncertainty.
Calculations with Significant Figures
Data processing involves the mathematical manipulation of data values recorded in the laboratory. When doing calculations with measurements, significant figures must be considered when writing a final solution. In general, the more significant figures, the more precise a measurement is. A calculation cannot make a result more precise so the final solution can never be more precise than the least precise value used in the calculation.
In other words, significant figures tell you how to round off your final answer.
For addition and subtraction calculations
- Determine the place value of the last significant digit in each number added or subtracted.
- Identify the measurement with the last digit of the highest place value.
- Round the final answer to that place value.
Example: Add the following: 120 cm, 34 cm, 4.5 cm and 0.40 cm.
Step 1. Identify the last significant digit in each number. These are the digits underlined in the given numbers.
Step 2. Identify the digit with the highest place value. This is the “2” in 120 cm as it is in the 10’s column.
Step 3. Add the numbers to get 158.9 cm. This must be rounded to the 10’s column so the final answer is 160 cm.
For multiplication and division (and combined operation) calculations
- Determine the number of significant figures of all the measurements used in the calculation.
- Identify the measurement with the least number of significant figures.
- Round the final answer to that number of significant figures.
Example: What is the area of a rectangle measuring 7.5 cm x 2.25 cm?
Count significant figures: 7.5 cm has 2 S.F. & 2.25 has 3 S.F. Therefore, the final answer can only have 2 S.F.
A = L x W
A = 7.5 x 2.25
A = 16.875
A = 17 cm2 (correctly rounded to 2 S.F.)
The IBO’s expectation for complicated calculations is to apply the general rule that the final solution has the same number of significant figures as the least precise measurement.
Here is Practice Worksheet 2.3 on Calcuations with Significant Figures.