Orders of Magnitude
Study in science deals with both very large and very small numbers.
The order of magnitude of a number is the power of 10 that is closest to the numerical value of a measurement or quantity.
Order of magnitude is closely related to scientific notation (reviewed in the following section) as a change of one order of magnitude is the same as multiplying or dividing a number by 10. For example 3452 is one order of magnitude larger than 345.2 and two orders of magnitude larger than 34.52.
The difference between two quantities in orders of magnitude is basically the difference between the base 10 exponents of the two quantities when written in scientific notation. For example my mass 8.95 x 101 kg is roughly 1032 or 32 orders of magnitude larger than the mass of an electron (9.11 x 1031 kg). That is a big difference!
Order of magnitude is important when we consider the range of measurements possible in our physical world i.e.
Object |
Mass (g) |
Electron |
9.11 x 10-28 |
Proton |
1.67 x 10-24 |
a molecule of CO2 |
7.30 x 10-23 |
mole of Carbon atoms |
1.20 x 101 |
person |
7.20 x 104 |
Moon |
7.35 x 1025 |
Order of magnitude is useful when approaching problems. Science involves numbers (lots of them) and frequent calculations. When you solve a problem, you can estimate your answer to the nearest power of 10, and compare to the solution you have obtained. If the solution differs by a couple of orders of magnitude you have probably made an error (using the calculator, or perhaps a unit conversion mistake or omission).
Careless mistakes!?!?!?
Example number 1
Suppose you just calculated the density of vegetable oil from lab data to be 9250 g/mL. How do you know this is wrong? What is the most likely error? What is the correct answer?
Example number 2
I just calculated that a mole of glucose (C6H12O6) has a mass of 1.80 x 1023 grams. Is this value realistic?
Example number 3
Check out this answer to a past physics exam question:
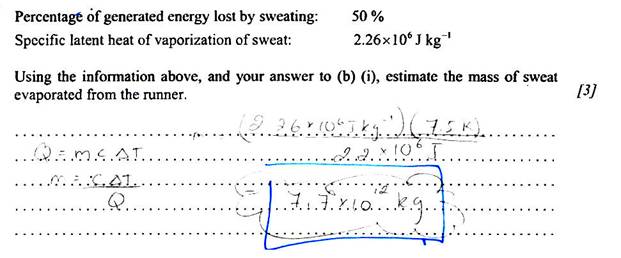
Clearly 7.7 × 1012 kg is unreasonable unless this person actually sweat enough water to fill 20 million swimming pools!
Expressing Orders of Magnitude: Scientific Notation
You will no doubt realize that it is convenient to express your answer in scientific notation (ie. it is a really big number). Frequently in science, very large and very small numbers are expressed in scientific notation. Scientific notation is used when expressing numbers to the correct number of significant figures and can also make unit conversions quite easy.
You must be comfortable with scientific notation and recognizing powers of 10. You must be able to enter these numbers in your calculator correctly if you are to succeed in science courses.
The general form for scientific notation is:
M x 10N
Where 1 < M < 10 and N is an integer.
Here is Practice Worksheet 1.1 on scientific notation and orders of magnitude.
The SI System of Measurement
- SI units (Le Systeme International d’Unites) – or more commonly called the metric system.
- Adopted for scientific research in 1960 worldwide, used as general measurement in most countries (USA and UK excepted)
Fundamental SI Units
- cannot be measured in simpler form
- you must know these basic quantities (and generally you need to convert them to solve problems)
Quantity |
Fundamental |
SI symbol |
Length |
Meter |
m |
Mass |
Kilogram or Gram |
kg or g |
Time |
Second |
s |
Electric Current |
Ampere |
A |
Absolute Temperature |
Kelvin |
K |
Amount of Substance |
Mole |
mol |
- some quantities are based on the measurement of two or more fundamental quantities – units for these are called derived units - a common example would be concentration (mol∙dm-3)
- note that the IBO accepted format for concentration is mol∙dm-3, not mol/dm3 or even mol/L . This applies to all derived units. (Get used to it!)
- the fundamental SI units are based on standards ex. the standard kilogram is a platinum-iridium cylinder kept in France, a standard second is the time for 9 192 631 770 vibrations of a cesium atom.
SI Unit Prefixes
Previously, the fundamental SI units were discussed. Much like scientific notation, larger and smaller SI units are created by multiplying the value of the base unit by factors of 10 (or 10-1). For example, the SI prefix “kilo” means multiplied by 103. Therefore 1 kilometer equals 103 or 1000 meters.
Common Prefixes used with SI Units |
|||
Prefix |
Symbol |
Meaning |
Order of Magnitude |
giga- |
G |
1 000 000 000 |
109 |
mega- |
M |
1 000 000 |
106 |
kilo- |
k |
1 000 |
103 |
hecto- |
h |
100 |
102 |
deka- |
da |
10 |
101 |
|
base unit |
1 |
100 |
deci- |
d |
0.1 |
10-1 |
centi- |
c |
0.01 |
10-2 |
milli- |
m |
0.001 |
10-3 |
micro- |
m |
0.000 001 |
10-6 |
nano- |
n |
0.000 000 001 |
10-9 |
Not all prefixes are commonly used. More detailed tables can often be found in your textbook or data booklets.
Note that there are many different ways a measurement can be expressed.
125 000 meters could be expressed as:
- 125 km
- 0.125 Mm
- 1.25 × 105
ALWAYS PAY ATTENTION TO UNITS!
Calculations in science involve the use of formulae and mathematical constants. Formulae often involve variables representing several different measured quantities. It is common practice in science that standard units are implied for the variables in the formulae. For example:
- most distances are measured in meters
- most masses are measured in kilograms (in physics) or in grams (in biology and chemistry)
- most time periods are measured in seconds.
Quantities measured in other units must first be converted
before a correct solution can be obtained.
Quite often, this can be simply accomplished by applying orders of magnitude. For example, the quantity 636 nm can be easily converted to meters and entered directly into the calculator as 636 × 10-9 m. (the 10-9 replaces the nano)
Here is Practice Worksheet 1.2 on the SI system and prefixes.