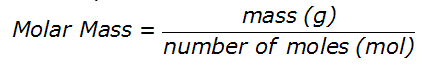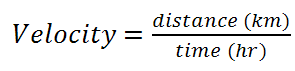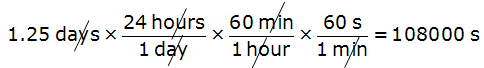Mathematical Skills
Applying mathematics is a critical aspect of problem solving in science. Many textbooks provide excellent mathematical review sections in the Appendices. Use these to refresh your knowledge or as a quick reference. The basic math skills required for IB science courses include the following:
Arithmetic and Computation
- Make calculations involving addition, subtraction, multiplication and division.
- Recognize and use expressions in decimal and standard form (scientific notation). Make calculations involving numbers in scientific notation.
- Use calculators to evaluate exponents; reciprocals; roots; logarithms to base 10; logarithms to base e; powers; arithmetic means; degrees; radians; quadratic equations; natural sine, cosine and tangent functions and their inverse.
*Note that some of these functions are only used in IB Physics. - Express fractions as percentages and vice versa.
Algebra
- Change the subject of an equation by manipulation of the terms including integer and fractional indices and square roots.
- Solve simple algebraic equations, and simultaneous linear equations involving two variables.
- Substitute numerical values into algebraic equations.
- Comprehend the meanings of (and use) the symbols
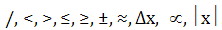
*Note that some of these symbols are only used in IB Physics.
Geometry and Trigonometry
*Note that all these are only used in IB Physics.
- Calculate areas of right-angled and isosceles triangles, circumferences and areas of circles, volumes of rectangular blocks, cylinders and spheres, and surface areas of rectangular blocks, cylinders and spheres. Relevant formulas need not be recalled.
- Use Pythagoras’ theorem, similarity of triangles and recall that the angles of a triangle add up to 180° (and of a rectangle, 360°).
- Understand the relationship between degrees and radians, and translate from one to the other.
- Recall the small angle approximations (tan theta = sin theta for small angles).
It is also extremely important that you are familiar with and know how to use your calculator efficiently and correctly. You do not learn how to solve problems by watching the teacher do them. To be successful, you must attack each problem aggressively and not be afraid to make mistakes.
BRING A CALCULATOR TO EVERY CLASS AND MAKE SURE YOU USE IT TO OBTAIN THE CORRECT SOLUTION FOR EVERY EXAMPLE PROBLEM WE DO!
Here is Practice Worksheet 4.1 on Math and Calculator Skills.
Dimensional Analysis
*Note that this technique is used primarily in Chemistry and Physics
The dimension of a measurement refers to its unit. Dimensional analysis means exactly that: pay attention to units! This simple example shows the benefit of dimensional analysis.
We know that molar mass is measured in grams per mole (g∙mol-1). Therefore the units give us the formula, namely:
We know that velocity is measured in km/hr or km∙hr-1. Therefore the units give us the formula, namely:
A good knowledge of the units you're using can help you figure out how to approach a science problem.
If you look at the units provided within the problem, you can figure out some ways that those units relate to each other and, in turn, this might give you a hint as to what you need to do to solve the problem. This process is known as dimensional analysis.
A Simple Physics Example:
Consider a basic problem that a student might get right after starting physics. A car moves a distance of 10 meters and a time of 60 seconds and you have to find the average velocity, but you don’t remember the equation to calculate velocity. Don't panic.
If you know your units, you know that velocity is measured in SI units of m/s or m*s-1. This means that there is a length (in meters) divided by a time (in seconds). You have a length and you have a time, so you're good to go.v = meters/seconds
= 10 m /60 s
= 0.17 m/s or 0.17 m*s-1
A Simple Chemistry example:
Often in chemistry, you are asked to find out a mole amount when learning stoichiometry. Consider the following question:
How many molecules of H2O are there in 0.5 moles of H2O?
First, set up the problem:
What am I being asked? Number of molecules (molecules is my unit)
What do I know? I have 0.5 moles H2O (moles is my unit)
I also know that there is a relationship between the units of moles and molecules – this is Avogadro’s Number! This number can be expressed in two ways – both equally true:
So the question is…do I divide by 6.02 x 1023 molecules or multiply? If you use dimensional analysis, it all takes care of itself! Basically, you want all of your units to cancel out mathematically, leaving you with only the unit of your answer!
Set up your equation like this:
_____________ x _______________ = molecules
The UNIT of molecules is a numerator (on the top!) and that’s what you’re looking for. Now organize all of your information, so that ALL OTHER UNITS CANCEL, EXCEPT FOR THE ONE YOU WANT. In this case molecules.
So essentially, you multiplied 0.5 moles by Avagadro’s number to get the answer. What would have happened with the units had you divided?
so just by looking at the units, you can see that the answer doesn’t make sense. You know you’ve done something wrong somewhere!
A More Complex Chemistry Example:
How many atoms of hydrogen are there in 0.5 moles of H2O?
see how all of the other units cancel out other than “atoms of H”? this is why it’s important to be very specific with units in this case!
A More Complex Physics Example:
How many moles of oxygen gas are there in 2.3 L of the gas at standard temperature and pressure?
From Avagadro’s Law, you know that there are 22.4 L of gas per mole of any gas, or 22.4 dm3*mol-1 at standard temperature and pressure.
Set up your problem…you want moles on the top!
Factor Label Method
Factor label method is a technique utilizing dimensional analysis to ensure the math is correct.
A conversion factor is an equality statement between two units. For example 1 m = 100 cm and 1 hour = 60 min are both conversion factors .
Factor-label method involves multiplying the value to be converted by appropriate conversion factors written as fractions (which have a value of 1). When arranging conversion factors as fractions, the numerator and denominator are determined by what unit is desired in the final answer and by what unit needs to be cancelled.
Example 1: Convert 4.5 m to cm.
Step 1. Determine the required conversion factor: 1 m = 100 cm
Step 2. Multiply 4.5 m by the conversion factor written as a fraction.
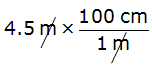
“m” cancels leaving “cm” and the answer 450 cmYou may need to multiply by a series of factors to get the conversion you want.
Example 2: Convert 1.25 days to seconds
In this example, days are converted to hours, then hours to minutes, and finally minutes to seconds to get the answer.
Note that significant figure rules do apply to unit conversions. The final answer should have the same number of significant figures as the original value.
Here is Practice Worksheet 4.2 on Unit Conversions.